A question for you: do we measure mass or weight on a laboratory balance? As we tell our students, weight and mass are not synonymous. Weight is the force of gravity acting on an object and varies depending on the object’s location. Mass, on the other hand, is the amount of matter in an object and remains constant regardless of location. So, which do we measure with a laboratory balance?
When we use a laboratory balance, we first calibrate it with certified standards that have a known mass. The balance then compares the force of gravity on our sample to the force of gravity on the mass standard. Since the standard’s mass is known, people typically say the balance measures the mass of our sample.
Indeed, a quick search online supports this consensus:
“A laboratory balance measures mass, not weight. Weight is the force exerted on an object due to gravity, while mass is the amount of matter in an object. The balance measures the mass of an object by comparing it to a known mass.” (https://www.studocu.com/ph/messages/question/3038573/does-a-laboratory-balance-measure-weight-or-mass-explain)
“On an analytical balance, we measure mass. Analytical balances are designed to provide highly accurate measurements of mass … in laboratory settings, especially with analytical balances, the term “weight” is often used colloquially, but the actual measurement being taken is mass.” (https://www.quora.com/Do-we-take-mass-or-weight-on-an-analytical-balance)
“In simple terms, a balances measure mass, while scales measure weight. So if you bring a balance and a scale to the moon, the balance will accurately give you the mass of, say, a moon rock, whereas the scale will be affected by the gravity.” (https://adamequipment.com/content/post/how-do-balances-and-scales-work#:~:text=In%20simple%20terms%2C%20a%20balances,mass%20against%20a%20known%20mass.)
Case closed, right? Well, not quite
Mettler Toledo, a respected instrument manufacturer, uses the term “weighing” for what we do in the lab. Mettler’s educational booklets do not use the term “massing,” nor do they say that we find the mass of a material using a lab balance. They say that we find its weight. Why is their terminology different?
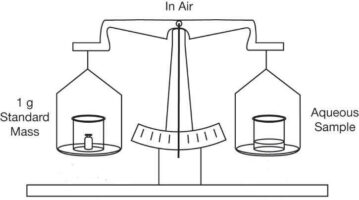
Mettler’s literature explains that the major force detected by a balance is, indeed, the force of gravity pulling down on the sample or standard. However, there is also a slight buoyant force from the air. The air around a sample or standard gives it a bit of lift and makes it appear lighter than it really is. This means that if the same object is weighed in air and in a vacuum, the object will be slightly heavier in the vacuum, as illustrated in the figure below. This is the principle of buoyancy: Any object will experience a loss in weight equal to the weight of the medium it displaces. Buoyancy is why helium balloons rise in the air.
Buoyancy force is proportional to the volume of air displaced, which depends on the object’s density. For example:
- A 1 g metal mass standard (high density) displaces less air than a 1 g sample of a powder (lower density).
- As a result, the powder experiences more buoyant force than the metal standard, causing a slight discrepancy between its mass and its measured weight. This is called buoyancy error and is small enough that it is ignored in most biotechnology settings.
The Effect of Buoyancy. a. A sample is exactly balanced against a 1 g mass standard in air. Although the sample and the standard have the same weight in air, they do not have the same mass. b. In a vacuum, the beam will tilt to the side with the sample.
Out of curiosity, I once approached a laboratory balance salesperson on a convention floor and asked him if anyone ever corrects for air buoyancy in the lab. He laughed and said that he had been hired to do exactly that in a drug-related case where the charge of felony versus misdemeanor hinged on the result of his analysis. That was interesting!
As a final note, if you look at the ASTM standard E1154, Standard Specification for Piston or Plunger Operated Volumetric Apparatus, you will find that there is a correction for buoyancy error built into a reference table that is used by meteorologists who calibrate micropipettes.
So, given all this, what’s your verdict – do we measure mass or weight in the laboratory?
You can check out Mettler’s excellent free educational resources, beginning with this one: Weighing the Right Way with Laboratory Balances. )